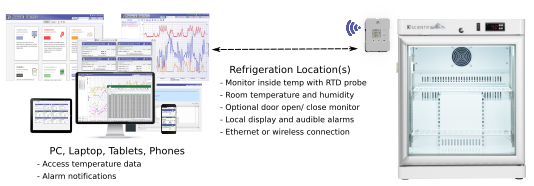
Temperature monitoring solutions for vaccine storage in refrigeration systems.
Paragon Robotics' monitoring and data logging systems provide real-time, 24/7 monitoring with programmable alarms, alert notifications and logged data to meet specific government regulations or customer requirements. Designed to meet FDA 21CFR Part 11 and CDC, VFC, and pharmaceutical regulations for auditing and reporting requirements. Our modular design incorporates accurate RTD temperature probes and simplifies sensor calibration and NIST certification.
- Wireless, Ethernet and USB connectivity options
- High resolution display and audible alarms
- Free cloud service for remote access and alarm notifications
- Built-in ambient temperature and humidity sensors
- Additional measurement capability through expansion ports and DB modules
- Broad range of add-on sensor modules available
- Software dashboards, charting analysis, data exports and reports
- Enterprise software upgrades for multi-user system with administrative options, auditing features, and custom reports
Our modular design approach allows each unit to be customized around the application or capable of future expansion as monitoring requirements expand. Our flexible, scalable, and customizable solutions will efficiently meet your most demanding applications.
Maintain operating temperatures for your vaccines, raw materials, and supplies across all your cooling systems and storage areas.
Monitor and record temperature, humidity, light, particulate, and carbon dioxide to ensure an optimal environment for product quality assurance.
- Centers for Disease Control/ Prevention (CDC)
- Vaccines For Children (VFC) program
- Food and Drug Administration (FDA)
- Pharmaceutical Boards.
All of our monitoring systems include our software apps with sensor dashboards, logging, charting NS analysis, data export and import capabilities, and individual sensor calibration. Our Enterprise software provides a multi-user environment with administrative privileges and user auditing features to meet compliance requirements.
- Optimize vaccine and medicinal storage conditions with continuous temperature monitoring.
- Monitor access and record door openings and closings
- Reduce labor-intensive reporting and documentation with automated 24/7 monitoring system and compliant software.
- Immediate email and/or text notification of cooling failures.
- Automate operations and improve work efficiency
Featured Products








Enterprise software for regulatory compliance requirements
Our monitoring systems and Enterprise software are designed to meet core industry regulation requirements for electronic documentation and signature control in a multi-user environment.
- Multiple user with administrative priviledges

- Meets 21CFR Part 11, VFC, CDC, ISO 9001, quality and custom industry regulatory software requirements.
- Multiple users with individual user logins, privileges and auditing features
- System and alarm notifications via email and text messages
- Cloud access to real-time sensor data
- ReportCreator for auditing, regulatory reporting and customized quality controls
- MessageCenter with system notifications and alarm tracking
- Advanced security with encrypted data storage
- Building models and sensor location layouts

Choose from a broad range of measurements including temperature, humidity, power, light, occupancy, moisture, pressure, air quality, CO2, particulate, or flow, as well as compatibility with any instrumentation or sensor output. Available sensor devices and external sensors are cost-effective and easy to install.
After initial setup and installation, the monitoring systems easily scales with additional sensors as your requirements expand. Systems are also compatible with any external sensors or existing instrumentation with VDC or mA outputs.
With battery operation and a large data storage capacity, you can quickly install your system and start running hassle-free. Our low-cost monitoring systems and data loggers provide fast installation and are the ideal choice for your monitoring, logging, and automation applications.





